
-
Vladislav Panin
- Professor, Biochemistry and Biophysics
- Focus Area: Therapeutics, Enzymology, Gene Regulation, Membrane Function, and Metabolism.
- Office:
- BICH 341A
- Email:
- vlad.panin@ag.tamu.edu
- Phone:
- 979-458-4630
Education
- Undergraduate Education
- B.S., M.S. - 1987
- Graduate Education
- Ph.D. - 1990
- Postdoc. - 2001
Areas of Expertise
- Therapeutics
- Enzymology
- Gene Regulation
- Membrane Function
- Metabolism
Professional Summary
We study protein glycosylation in different biological contexts, including physiology, development, and disease. It has been long recognized that glycans play essential roles in animal organisms, while defects in glycosylation are associated with numerous diseases and abnormalities, from cancer to brain malformation and defects in neurophysiology. However, the complexity of glycosylation and limitations of in vivo approaches impede research in mammals. We are using advantages of Drosophila model, including its powerful arsenal of genetic and in vivo approaches, to elucidate pathomechanisms underlying human diseases caused by glycosylation defects.
Pathomechanisms of abnormal protein O-Mannosylation
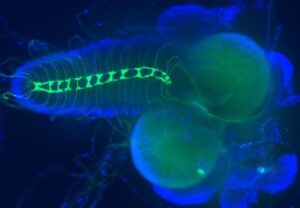
POM mutations cause defects of sensory axon connections (green) in the larval brain (blue), which results in abnormal muscle contractions and body torsion phenotype (Monagas-Valentin, et al. 2023).
Our projects are focused on the mechanisms of protein O-mannosylation (POM) in neural development. Defects in POM have been implicated in severe disorders, such as muscular dystrophy, and lissencephaly. Using Drosophila as a model system, we discovered that POM is essential for the wiring of sensory axons and the neural control of muscle contractions. This project opened new avenues of research on new POM substrates and their functions in the regulation of neural connectivity. We currently employ a combination of genetic and biochemical approaches to identify novel functional targets of POM in the nervous system and characterize new enzymes involved in the POM pathway.
Pedro Monagas-Valentin, Robert Bridger, Ishita Chandel, Melissa Koff, Boris Novikov, Patrick Schroeder, Lance Wells, Vladislav Panin (2023) Protein Tyrosine Phosphatase 69D is a substrate of Protein O-Mannosyltransferases 1-2 that is required for the wiring of sensory axons in Drosophila. J Biol Chem. 299(3):102890. doi: 10.1016/j.jbc.2023.102890.
Regulation of neural transmission by sialylation
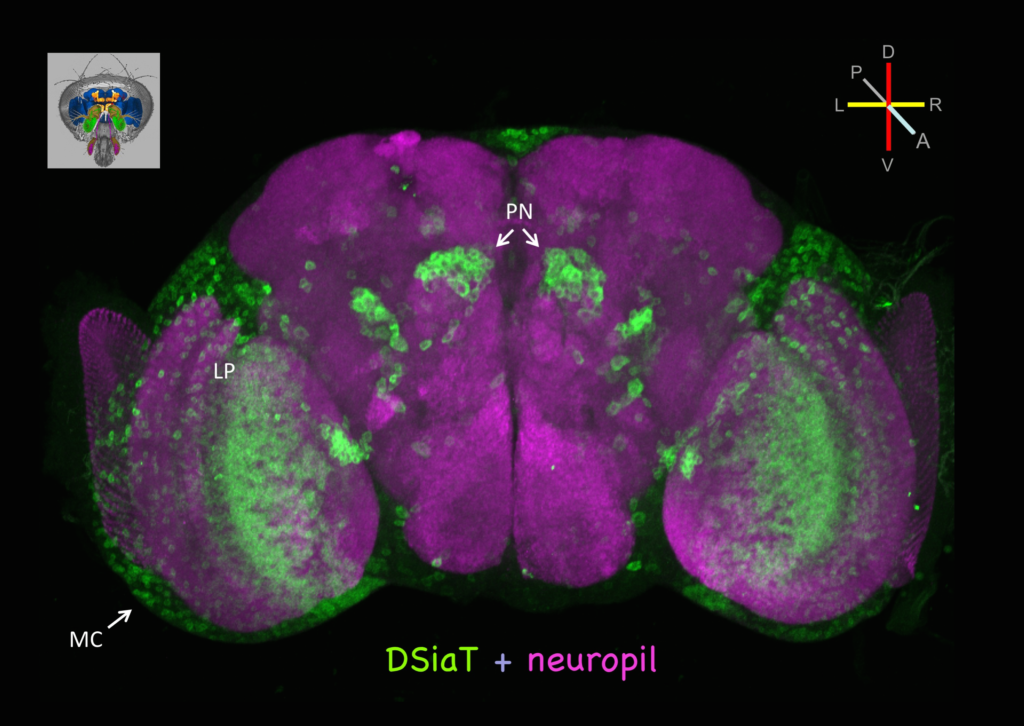
DSiaT is expressed in numerous cells in the adult brain, including neurons of the optic lobes (MC, medulla cortex; LP, lobula plate) and the olfactory system (PN, projection neurons).
J. Neurosci. 33, 12306–12315 (2013).
We investigate the role of neural sialylation in the regulation of neural development, excitability and transmission. We identified and characterized several key sialylation enzymes in Drosophila, including Drosophila sialyltransferase (DSiaT) and CMP-sialic acid synthetase (CSAS). Genetic inactivation of the sialylation pathway in vivo unveiled prominent roles of sialylated N-glycans in the regulation of the nervous system. Sialylation mutants have significantly reduced life span, locomotor abnormalities, and temperature-sensitive paralysis phenotype. We found that sialylation affects synaptic growth and the function of voltage-gated channels. Our studies established a model system to elucidate how this prominent glycosylation pathway controls neural functions. We currently investigate the molecular and cellular mechanisms that underlie the sialylation-mediated control of neural functions during development and aging.
Hilary Scott, Boris Novikov, Berrak Ugur, Brooke Allen, Ilya Mertsalov, Pedro Monagas-Valentin, Melissa Koff, Sarah Baas Robinson, Kazuhiro Aoki, Raisa Veizaj, Dirk Lefeber, Michael Tiemeyer, Hugo Bellen, Vlad Panin (2023) Glia-neuron coupling via a bipartite sialylation pathway promotes neural transmission and stress tolerance in Drosophila. eLife, 2023 Mar 22;12:e78280. doi: 10.7554/eLife.78280
Publications
- View publications on PubMed